Draw A Lewis Structure For Hcn
Draw A Lewis Structure For Hcn - Put one electron pair in each bond 4. Establish a general procedure for drawing lewis structures. With the lewis structure for hcn you’ll need to share more than one pair of electrons between the carbon and the nitrogen atoms. #1 first draw a rough sketch. Hcn lewis dot structure by counting valence electrons on the carbon and nitrogen atom. Here we have to find the valence electrons of all three atoms, hydrogen, carbon, and nitrogen.
Web to sketch the hcn lewis structure by following these instructions: 3.8k views 6 years ago chem. H + c + n =1 + 4 + 5 = 10. Add the valence electrons of all the molecules' atoms: Count the valence electrons you can use.
the chemical symbol for h cen is shown in black and white with an
= 4 + 1x4 = 8 valence electrons; Lewis structure of hcn for counting valence electrons around the terminal hydrogen atoms. Hcn lewis dot structure by counting valence electrons on the carbon and nitrogen atom. In hcn, the carbon atom forms a triple bond with the nitrogen atom. Web the lewis structure (lewis dot diagram) for hcn.
Hcn Lewis Structure How To Draw The Lewis Structure For Hcn Youtube
In order to draw the lewis structure of hcn, first of all you have to find the total number of valence electrons present in the hcn molecule. Web drawing lewis structures for molecules with one central atom: Draw the lewis structure for hcn. Put the least electronegative atom c in the middle with h and cl on either side. You.
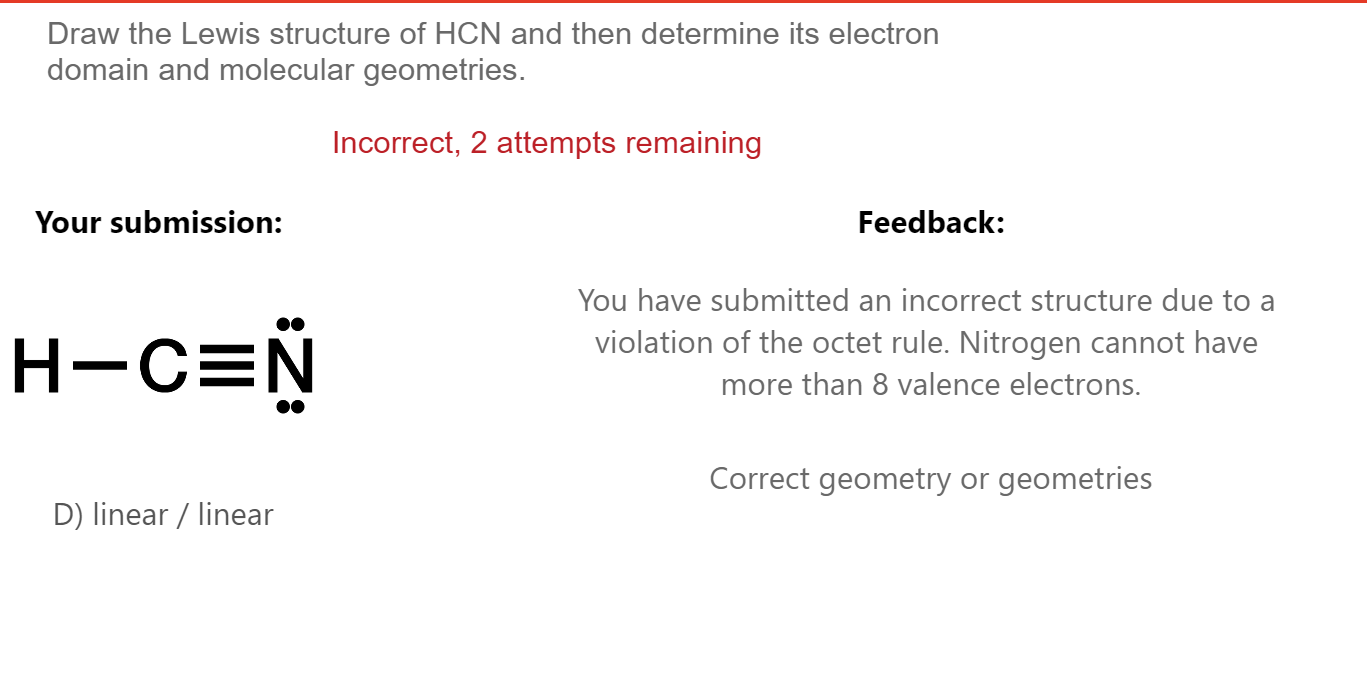
Solved Draw the Lewis structure of HCN and then determine
Hydrogen has 1 valence electron, carbon has 4 valence electrons, and nitrogen has 5 valence electrons. Calculate the total number of valence electrons. In hcn, the carbon atom forms a triple bond with the nitrogen atom. Web to draw the lewis diagram for hcn, we need to consider the valence electrons of each atom. You nave to put a triple.
35 Lewis Dot Diagram For Hcn Wiring Diagram Database
Pcl 3 has 5 valence electros in p and 7 in each of the three cl: The hydrogen atom is bonded to the carbon atom, and the nitrogen atom is bonded to the carbon atom via a triple bond. #3 calculate and mark formal charges on the atoms, if required. Web drawing the lewis structure for hcn. Web drawing lewis.
[Solved] what is the lewis structure, electron geometry, molecular
= 5 + 7x3 = 26 valence electrons In the lewis diagram for hcn, we represent the valence electrons of each atom using dots. Web learn to draw the lewis structure of hcn & understand molecular geometry, shape, & polarity about the same by reading this article. Web the lewis structure of hcn shows that the carbon atom is the.
Draw A Lewis Structure For Hcn - H + c + n =1 + 4 + 5 = 10. #2 mark lone pairs on the atoms. = 4 + 1x4 = 8 valence electrons; Here we have to find the valence electrons of all three atoms, hydrogen, carbon, and nitrogen. Web draw lewis structures depicting the bonding in simple molecules. Web how to draw lewis structure for hcn?
Web does this molecule | chegg.com. Add the valence electrons of all the molecules' atoms: Web to draw the lewis diagram for hcn, we need to consider the valence electrons of each atom. Fill outer atoms with electrons 5. Lewis structure of hcn for counting valence electrons around the terminal hydrogen atoms.
The Hydrogen Atom Is Bonded To The Carbon Atom, And The Nitrogen Atom Is Bonded To The Carbon Atom Via A Triple Bond.
Web drawing the lewis structure for hcn. Put least electronegative atom in centre 3. Hydrogen has 1 valence electron, carbon has 4 valence electrons, and nitrogen has 5 valence electrons. Add the valence electrons of all the molecules' atoms:
= 4 + 1X4 = 8 Valence Electrons;
3.8k views 6 years ago chem. Here, the given molecule is hcn. Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. Here is a little flow chart of how we are going to do this:
= 5 + 7X3 = 26 Valence Electrons
Web 0:00 / 2:14. Lewis structure of hcn for counting valence electrons around the terminal hydrogen atoms. Add these electrons to give every atom an octet. You nave to put a triple bond between c and n.
Fill Outer Atoms With Electrons 5.
Web does this molecule | chegg.com. #2 mark lone pairs on the atoms. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. How to draw lewis structure for hcn?