Draw The Predominant Form Of Histidine At Ph 0
Draw The Predominant Form Of Histidine At Ph 0 - Step by step solved in 3 steps with 1 images. Web | science | chemistry. Web chemistry questions and answers. Trending now this is a popular solution! Check out a sample q&a here. At a ph of 2.9, which is below the pka value of the histidine side chain (6.1), the histidine molecule will predominantly be in a fully protonated form.
This is because the ph scale measures the concentration of hydrogen ions in a solution, and at low ph, there is an abundance of h⁺. Check out a sample q&a here. Web there are only one or two dominant species present at a particular ph. After completing this section, you should be able to. Step by step solved in 3 steps with 2 images.
SOLVEDDraw the most predominant form of histidine at its isoelectric
This problem has been solved! Web | science | chemistry. Histidine will be deprotenated at 7.4 ph ( since 7.4> 6.5) and it will be acidic, it will rain negative charge and will be acidic; (b) draw the structures of the predominant ionization state of histidine at ph 1, 4, 8, and 12. At low ph, such as ph=0, histidine.
Solved Draw the predominant form of Lhistidine at pH=7.6.
At a ph ≈ (1.7+6.0)/2 = 3.9, 98 % is in the h2a+ form. The pka of the side chain is 6.1. At a ph of 2.9, which is below the pka value of the histidine side chain (6.1), the histidine molecule will predominantly be in a fully protonated form. Below ph = 1.7 (pka1) h3a2+ is the most abundant.
draw the predominant form of histidine at ph 0
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Draw histidine in its predominant form at ph = 10.0. At ph = 6.02 there is equal amounts of h2a+ and ha. Histidine is an essential amino acid.
Histidine Chemical Formula. Histidine 3D Realistic Chemical Molecular
Below ph = 1.7 (pka1) h3a2+ is the most abundant specie. Check out a sample q&a here. Here’s the best way to solve it. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Draw the predominant form of histidine at the following ph values.
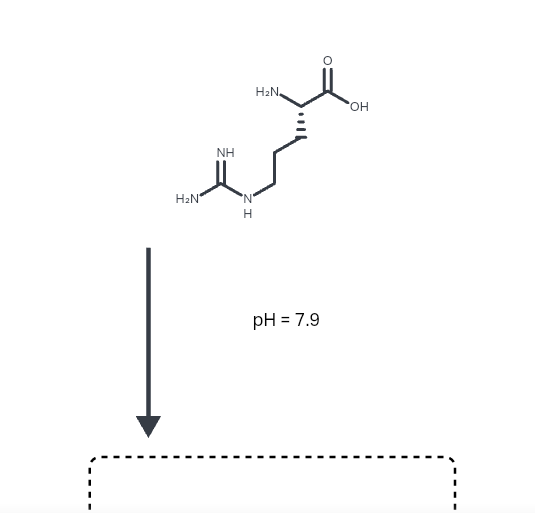
Solved Draw the predominant form of arginine at pH = 7.9.
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Step by step solved in 3 steps with 1 images. At ph 2 at ph 7.8 at ph 10 would you expect the amino acid tyrosine (a neutral amino acid) to be soluble at ph 8? Web ph = 0 ph = 7 ph.
Draw The Predominant Form Of Histidine At Ph 0 - April 22, 2022 by triyasha mondal. At low ph, such as ph=0, histidine exists primarily in its protonated form as h⁺. Here’s the best way to solve it. (b) draw the structures of the predominant ionization state of histidine at ph 1, 4, 8, and 12. Web if the answer to my question is yes, then why histidine is listed as basic amino acids then? So hissed adede or h i answer.
Draw the predominant form of histidine at ph=0. The pka of the side chain is 6.1. Describe, briefly, how a mixture of amino acids may be separated by paper electrophoresis. Draw the predominant form the histidine amino acids at physiological ph (7.4): (b) draw the structures of the predominant ionization state of histidine at ph 1, 4, 8, and 12.
Web | Science | Chemistry.
Draw histidine in its predominant form at ph = 10.0. Pka 3.86 nh2 o ho o oh glutamic acid pka 4.25 histidine nh2 o ho n+ n h h pka 6.04 nh2 o ho sh cysteine pka 8.35 lysine nh2 o ho nh3+ pka 10.79 nh2 o ho oh tyrosine pka 10.07 arginine nh2 o ho h n nh2 nh2+ pka 12.48. Web chemistry questions and answers. This problem has been solved!
Draw Histidine In Its Predominant Form At Ph = 2.0.
Histidine is fully protonated at ph 2.9, resulting in a positive charge on the imidazole ring in the side chain and the amino group, and a neutral carboxylic acid group. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web draw the predominant form of a given amino acid in a solution of known ph, given the isoelectric point of the amino acid. (a) in ph 5 b, in ph 10 c, in ph 10.9 d, what is the net charge of histidine at the ph 10.
At Low Ph, Such As Ph=0, Histidine Exists Primarily In Its Protonated Form As H⁺.
So hissed adede or h i answer. Note that the ionization state can be approximated by treating each ionizable group. The predominant form of histidine at ph=0 is: Web chemistry questions and answers.
Step By Step Solved In 3 Steps With 2 Images.
Check out a sample q&a here. Draw arginine in its predominant form at ph = 2.0. Keep in mind that the pk of an ionizable group is influenced by its microenvironment, so the pk values of amino acids may vary by several ph units when the amino acid is part of a folded polypeptide chain. At ph = 1.7 there is a equal amount of h3a2 + and h2a+.